
Back Koolstofmonoksied Afrikaans Monoxido de carbonio AN أحادي أكسيد الكربون Arabic Monóxidu de carbonu AST Karbon monoksid Azerbaijani کربون مونوکسید AZB Smalkē BAT-SMG Въглероден оксид Bulgarian कार्बन मोनोऑक्साइड Bihari কার্বন মনোক্সাইড Bengali/Bangla
| |||
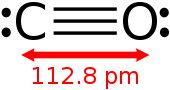
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Carbon monoxide
| |||
| Other names
Carbonic oxide gas
Carbon protoxide Oxide of carbon Protoxide of carbon Carbonous oxide Carbonous acid gas Carbon(II) oxide Breath of carbon Oxygenated carbon Carbate Carbonyl Water gas Hydrocarbon gas Fuel gas Rauchgas Carbonic inflammable air Heavy inflammable air White damp Fire Damp Powder Gas Illuminating gas Dowson gas Mond gas Power gas Producer gas Blast furnace gas Coal gas Phlogiston Car gas | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 3587264 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.010.118 | ||
| EC Number |
| ||
| 421 | |||
| KEGG | |||
| MeSH | Carbon+monoxide | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1016 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CO | |||
| Molar mass | 28.010 g·mol−1 | ||
| Appearance | Colorless | ||
| Odor | Odorless | ||
| Density |
| ||
| Melting point | −205.02 °C (−337.04 °F; 68.13 K) | ||
| Boiling point | −191.5 °C (−312.7 °F; 81.6 K) | ||
| 27.6 mg/L (25 °C) | |||
| Solubility | soluble in chloroform, acetic acid, ethyl acetate, ethanol, ammonium hydroxide, benzene | ||
Henry's law
constant (kH) |
1.04 atm·m3/mol | ||
| −9.8·10−6 cm3/mol | |||
Refractive index (nD)
|
1.0003364 | ||
| 0.122 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
29.1 J/(K·mol) | ||
Std molar
entropy (S⦵298) |
197.7 J/(K·mol) | ||
Std enthalpy of
formation (ΔfH⦵298) |
−110.5 kJ/mol | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−283.0 kJ/mol | ||
| Pharmacology | |||
| V04CX08 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Poisonous by inhalation[1] | ||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H220, H331, H360, H372, H420 | |||
| P201, P202, P210, P251, P260, P261, P264, P270, P281, P304+P340, P308+P313, P311, P314, P321, P377, P381, P403, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −191 °C (−311.8 °F; 82.1 K) | ||
| 609 °C (1,128 °F; 882 K) | |||
| Explosive limits | 12.5–74.2% | ||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration)
|
| ||
LCLo (lowest published)
|
| ||
| NIOSH (US health exposure limits):[1] | |||
PEL (Permissible)
|
TWA 50 ppm (55 mg/m3) | ||
REL (Recommended)
|
| ||
IDLH (Immediate danger)
|
1200 ppm | ||
| Safety data sheet (SDS) | ICSC 0023 | ||
| Related compounds | |||
Other anions
|
Carbon monosulfide | ||
Other cations
|
Silicon monoxide Germanium monoxide Tin(II) oxide Lead(II) oxide | ||
Related carbon oxides
|
Carbon dioxide Carbon suboxide Oxocarbons | ||
| Supplementary data page | |||
| Carbon monoxide (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Carbon monoxide (chemical formula CO) is a poisonous, flammable gas that is colorless, odorless, tasteless, and slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simplest carbon oxide. In coordination complexes, the carbon monoxide ligand is called carbonyl. It is a key ingredient in many processes in industrial chemistry.[5]
The most common source of carbon monoxide is the partial combustion of carbon-containing compounds. Numerous environmental and biological sources generate carbon monoxide. In industry, carbon monoxide is important in the production of many compounds, including drugs, fragrances, and fuels.[6] Upon emission into the atmosphere, carbon monoxide affects several processes that contribute to climate change.[7]
Carbon monoxide has important biological roles across phylogenetic kingdoms. It is produced by many organisms, including humans. In mammalian physiology, carbon monoxide is a classical example of hormesis where low concentrations serve as an endogenous neurotransmitter (gasotransmitter) and high concentrations are toxic resulting in carbon monoxide poisoning. It is isoelectronic with both cyanide anion CN− and molecular nitrogen N2.
- ^ a b NIOSH Pocket Guide to Chemical Hazards. "#0105". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Carbon monoxide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Richard, Pohanish (2012). Sittig's Handbook of Toxic and Hazardous Chemicals and Carcinogens (2 ed.). Elsevier. p. 572. ISBN 978-1-4377-7869-4. Retrieved 5 September 2015.
- ^ "Carbon Monoxide - CAMEO Chemicals". cameochemicals.noaa.gov. US NOAA Office of Response and Restoration.
- ^ Cite error: The named reference
Ullwas invoked but never defined (see the help page). - ^ Bierhals, Jürgen (2001). "Carbon Monoxide". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a05_203. ISBN 3527306730.
- ^ Voiland, Adam. "Fourteen years of carbon monoxide from MOPITT". Climate Change: Vital Signs of the Planet. Retrieved 2022-03-04.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search


